Vacteens: A Mobile Web App to Improve HPV Vaccine Uptake
Dr. W. Gill Woodall from Klein Buendel (KB) and the University of New Mexico and Jeanny Reither from KB presented findings from the Vacteens Project at the Eurogin International Multidisciplinary HPV Congress in Lisbon, Portugal, December 2-5, 2018. Eurogin is one of the most important conferences on Human Papillomavirus (HPV) infection and related cancers. It aims to raise the public health profile of HPV, increase the need for responsible health services, and examine the cost-effectiveness of risk-based screening to pave the way for the development of new strategies for the prevention of HPV-induced cancers.
The uptake of the HPV vaccine in the United States remains significantly below the Healthy People 2020 goal of 80% series completion, and this is particularly so for the young adolescent age range, when the immunogenic response to the vaccine is stronger. While a number of factors may account for this less than desirable vaccine uptake, parental concerns and misinformation about the efficacy and safety of HPV vaccine remain barriers to reaching public health vaccination goals. Physician and clinic-based interventions have shown some limited positive effect on vaccine uptake. However, parental barriers to HPV vaccination may ideally be addressed by digital interventions (in this case, smartphone applications) that are tailored to their concerns. Specifically, research indicates there is a great deal of: (1) confusion and uncertainty about HPV vaccine, and (2) concomitant misinformation about HPV vaccine, who it is meant for, and the conditions under which it is maximally effective.
With funding from the Patient Centered Outcomes Research Institute (PCORI) and the National Cancer Institute, Dr. Woodall’s team systematically developed a set of mobile web app tools to prompt the informed adoption of HPV vaccination. They used Diffusion of Innovations Theory and related research on Informed Decision Making to guide the iterative development of mobile apps for parents of young female and male adolescents.
Ms. Reither presented a poster that reported the results of developmental research and early trial findings from two smartphone web app projects — one focused on parents and adolescent girls (ages 11-14) and the other on parents and adolescent boys in the same age range. The objective of these investigations is to develop and evaluate a mobile web app to encourage HPV vaccination in New Mexico, an ethnically-diverse state. Current ongoing randomized controlled efficacy trials with parents and their adolescent children in New Mexico clinics provide data to determine the impact of these mobile web apps on informed decision making and uptake for the HPV vaccine.
Dr. Woodall participated in a panel session entitled “Uses of New Technologies in HPV Vaccine Behavioral Science Research.” He gave a presentation on the design, development, and testing of the innovative Vacteens web app. New technologies and social networking sites like this can be used to understand sources of information and misinformation about HPV vaccination, engage parents and youth, and encourage HPV vaccination.
The progress and initial results of these ongoing research efforts will have implications for reaching HPV vaccine uptake goals set by Healthy People 2020 in the United States. Mobile web-based interventions show promise for reaching HPV vaccine uptake goals. A mobile web app can make decision-making tools widely available on popular mobile platforms such as tablet computers and smartphones, as well as personal computers.
This research was funded by grants from PCORI and the National Cancer Institute (CA210125; Dr. W. Gill Woodall, Principal Investigator). Collaborators included Dr. Alberta Kong, Dr. Randall Starling, Dr. Lance Chilton, and Dr. Tamar Ginossar from the University of New Mexico; Dr. Greg Zimet from Indiana University; and Dr. David Buller and Jeanny Reither from KB.

Dr. W. Gill Woodall and Jeanny Reither from Klein Buendel

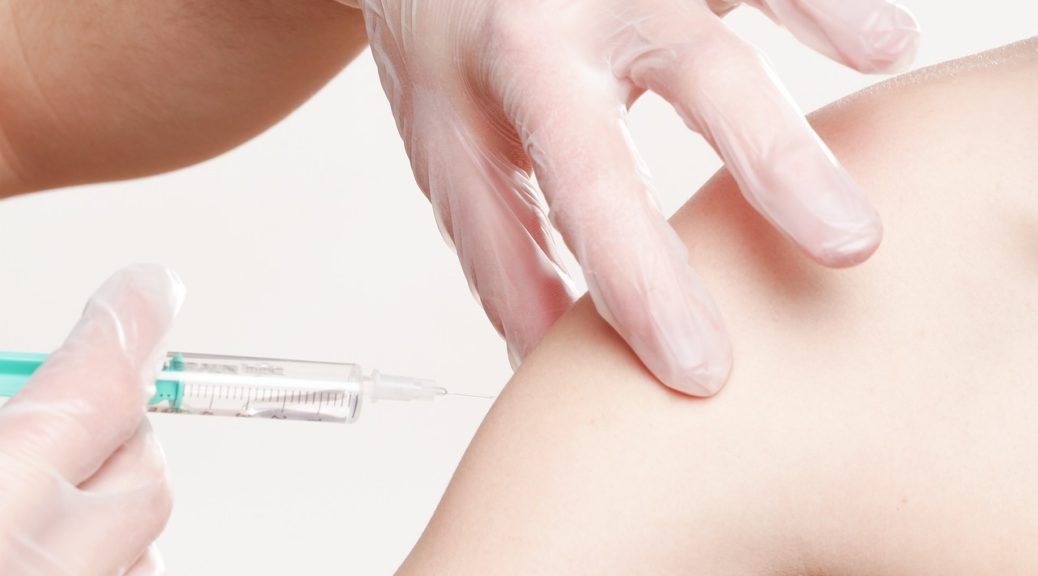

 Dr. Gregory Zimet is a Clinical Psychologist and Professor in the Department of Pediatrics at the Indiana University School of Medicine. Since he arrived there in 1993, he has guided an extensive, multi-faceted research program focused on attitudes about, and acceptance of, vaccines for the prevention of sexually-transmitted infections, including the human papillomavirus (HPV) vaccine.
Dr. Gregory Zimet is a Clinical Psychologist and Professor in the Department of Pediatrics at the Indiana University School of Medicine. Since he arrived there in 1993, he has guided an extensive, multi-faceted research program focused on attitudes about, and acceptance of, vaccines for the prevention of sexually-transmitted infections, including the human papillomavirus (HPV) vaccine.